Community Engagement, Communications, and Technology for Clinical Trials in Outbreak Settings: EBODAC Training Resource
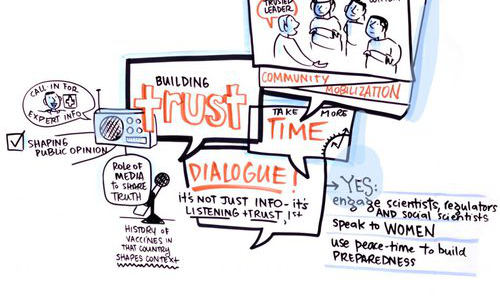
The EBODAC project (Ebola Vaccine Deployment, Acceptance and Compliance) is developing communications and engagement strategies and tools to support Ebola vaccine trials and to promote the acceptance and uptake of new Ebola vaccines. In February 2017, the EBODAC consortium - London School of Hygiene & Tropical Medicine (LSHTM), Janssen Pharmaceutica NV., World Vision, and Grameen Foundation - organised a symposium on this theme in Dakar, Senegal, which brought together experts from across Africa and beyond. The learnings from the symposium have been incorporated into a training resource that is available as an open-access tool for anybody who is interested in community engagement, communications, and enabling technologies for clinical trials in outbreak settings.
The EBODAC training resource includes the following sections:
- Introduction - describes the context, which was the 2014-2016 Ebola outbreak that devastated communities across Guinea, Liberia, and Sierra Leone, leading multiple international research groups to rapidly mobilise themselves and work to establish clinical trials of both candidate Ebola vaccines and treatments. The ethical, scientific, and logistical challenges of setting up rapid and robust clinical trials in such a context were significant, creating the risk that the sensitivities, anxieties, and realities of potential trial participants and communities risked being considered as an afterthought rather than placed at the heart of the research itself.
- Ethical Challenges - focuses on issues that can serve as potential challenges for communications and community engagement teams working on clinical trials, taking an example-based approach that is intended to be illustrative. Sub-topics:
- Prioritisation of Research
- Prioritisation of Participants
- Community Involvement
- Informed Consent
- Social Context - discusses one of the first tasks researchers encounter: that of establishing lines of communication with the community, which in turn entails identifying community leaders. Sub-topic:
- The Role of Social Scientists
- Community Engagement in Practice - explores this practical process, which demands creative problem solving. Sub-topics:
- "Top-Down" versus "Bottom-Up"
- Working with Those New to Clinical Research
- Identifying Sticking Points: The Example of Blood Theft
- Guidelines for Community Engagement
- Rumours - features, for example, a short documentary film (see below), made by the Vaccine Confidence Project as part of EBODAC, looking at the Ghana vaccine trial cancellations through interviews with key actors involved in the public controversy. Sub-topics:
- The Example of Ghana
- How to Manage Rumours
- Enabling Technologies - examines several technologies used to keep track of participant identity (biometrics, including fingerprint and iris scanning) and maintain communication and follow-up with participants (MOTECH communication platform). Sub-topics:
- ICTs [Information and Communication Technologies] in Support of Clinical Trials
- Overcoming Barriers
- Research
- Robust training
- Community engagement and outreach strategies
- Tailoring technologies to maximise participant engagement
- Overcoming the barriers of electrical power and mobile connectivity
- Collaboration with local and international partners
- Recommendations for Using Enabling Technologies in Clinical Trials
- Use previous learnings and experiences
- Need for flexibility
- Need for harmonisation
- Conclusion - "Moving forward, the lessons from this crisis need to be used to improve the planning and implementation of clinical trials in the future, both in routine settings and crises. As the Ebola epidemic demonstrated, the community's involvement in any crisis situation is absolutely critical to its success."
EBODAC website, June 11 2018. Illustration credit: Sam Bradd (Drawing Change)
- Log in to post comments












































