Did the New Italian Law on Mandatory Vaccines Affect Adverse Event Following Immunization's Reporting? A Pharmacovigilance Study in Southern Italy
University of Campania "Luigi Vanvitelli" (Scavone, Rafaniello, Brusco, Bertini, Sportiello, Rossi, Capuano); University of Naples Federico II (Menditto, Orlando); Regional Pharmaceutical Unit, Campania Region (Trama)
"...the continuous monitoring of vaccine safety and the fully implementation of vaccine–vigilance programs play a key role in achieving higher confidence in immunization programs and optimal vaccination coverage rate."
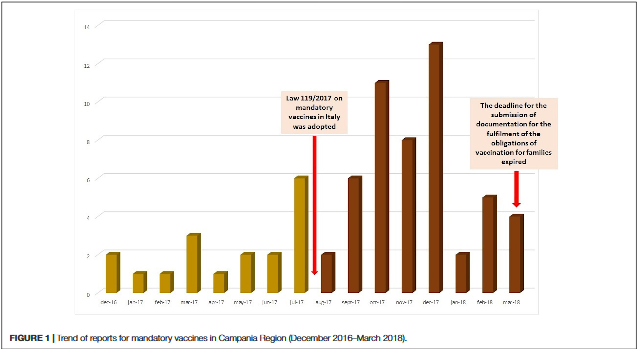
Vaccination coverage is far from optimal, especially in children, representing a growing concern in Italy. In order to address this problem, the Italian Ministry approved in July 2017 the Law 119/2017, which renders mandatory and free of charge 10 vaccinations for patients aged 0-16. Considering that the rate of adverse events following immunisation (AEFIs) reports may increase in response to media attention and increased public awareness, this article investigates the effects of the new Law 119/2017 on the spontaneous reporting of AEFIs into the in Campania region's Italian Pharmacovigilance database (Rete Nazionale di Farmacovigilanza - RNF). Through the RNF, physicians, other healthcare professionals, and patients/citizens can send reports of suspected adverse drug reactions (ADRs) and AEFIs using a standardised reporting form.
All AEFIs' reports regarding mandatory vaccines (H. influenzae type B, measles, mumps, rubella, varicella, pertussis, diphtheria, tetanus, polio, and hepatitis B), as suspected and validated by the Campania Pharmacovigilance Regional Center from December 1 2016 to March 31 2018, were selected.
There were 69 reports covering 179 AEFIs related to mandatory vaccines. The highest number of reports came from patients/citizens. A substantial increase in AEFIs reporting was observed after the adoption of the Law 119/2017 (18 before the adoption of the law vs. 51 after) Out of 69 reports, 62% reported AEFIs that were considered as not serious, and 78% had a favourable outcome. Out of 179 AEFIs, more than half referred to the following system organ class (SOC): "general disorders and administration site conditions", "nervous system disorders", and "psychiatric disorders".
According to the Italian Medicines Agency (AIFA), the increase in the number of AEFI reports coincided with the beginning of the parliamentary debate on the decree that preceded the law and the fully activation of Vigifarmaco, a new web reporting and transit platform to the RNF established by AIFA in 2015. Indeed, in all Italian regions, AEFI reports rose from 35% in 2016 to 54% in 2017, and the overall reporting rate for vaccines increased from 7.9/100,000 inhabitants in 2016 to 11.1/100,000 in 2017. The increase in AEFI reports could also be related to the intense debate that has followed the new law, which was mainly carried out by the so-called Free-Vaxxers group.
The researchers suggest that another explanation for the increase could be "the failure of risk communication campaign on the safety of vaccines. As a matter of fact, since citizens' decision on immunization must depend on regulatory agencies', health ministries', and clinicians' choices, a huge attention should be paid to their education and information on disease risk and prevention of risk, through an open dialog based on empathy, respect, and transparency....In this context, also the management of real world studies...will help to improve the knowledge on the safety profile of drugs and vaccines."
As the researchers conclude: "Along with the rapid increase in the coverage for compulsory vaccines registered in Campania Region after the adoption of the Law, there was also an increase in AEFIs' reporting. In our opinion, reasons for this are no doubt numerous, including the increase in vaccination coverage, the huge media attention that has followed the new Law, the increased citizens' awareness of AEFIs in children as well as a greater empowerment and involvement of patients in pharmacovigilance activities. Despite those effects on AEFIs' reporting, there is no doubt about the enormous positive consequences of the Law 119/2017 for public health..."
Frontiers in Pharmacology. 2018; 9: 1003. doi: 10.3389/fphar.2018.01003.
- Log in to post comments












































