A Scoping Review of Active, Participant-Centred, Digital Adverse Events Following Immunization (AEFI) Surveillance: A Canadian Immunization Research Network Study
Ottawa Hospital Research Institute (Psihogios, Bota, Mithani, Zhu, Wilson); University of British Columbia (Greyson, Bettinger); Children's Hospital of Eastern Ontario (CHEO) Research Institute (Fung, Fell); Public Health Ontario (Wilson); University of Toronto (Wilson); ICES (Wilson); University of Ottawa (Fell, Wilson); Dalhousie University (Top); Bruyère Research Institute (Wilson)
"Vaccine safety, and the assessment of surveillance approaches through formal investigation, is a critical aspect of immunization programs..."
Post-licensure adverse events following immunisation (AEFI) surveillance helps maintain community trust and promotes public uptake of vaccines. Participant-centred active surveillance involves the self-reporting of health events or health status by individuals following vaccination. This scoping review considers digital AEFI surveillance, which uses electronic technology for capturing and processing data. The review's objectives are to: (i) map the research landscape on digital systems used for active, participant-centred, AEFI surveillance and (ii) describe their core components.
Participant-centred reporting is different from, but can complement, passive AEFI surveillance. Passive surveillance is recommended by the World Health Organization (WHO), but it has its limitations, such as underreporting and the potential for reporting bias. With active participant-centred surveillance, response rates can be calculated to gauge degree of reporting, questionnaires can be used to collect specific data to improve both the quality and completeness of reports, and contextual information can be collected through follow-up to facilitate the identification of new signals in a timely manner. Serious adverse event follow up can be built into the system with relatively low resource requirements, and the incorporation of control groups can inform baseline incidence rates.
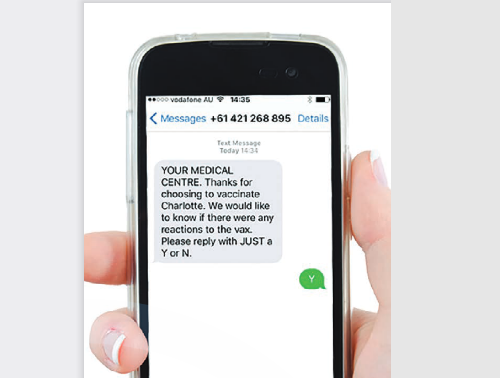
Twenty-seven publications from the review, which covered the period January 1 2000 to January 28 2021, met inclusion. The digital-based systems identified in the literature landscape included SmartVax (Australia), Vaxtracker (Australia), Stimulated Telephone Assisted Rapid Safety Surveillance (STARSS) (Australia), Canadian National Vaccine Safety (CANVAS) Network (Canada), FAST-Mum (Australia), FAST-Health (Australia), Influenzanet (Netherlands), Lareb Intensive Monitoring System (Netherlands), Voxiva (United States - US), TeleWatch (US), and the Integrated Vaccine Surveillance System (IVSS) (US). Table 3 in the paper offers a detailed description of the systems, including population under surveillance, communication methods, AEFI reporting methods, vaccinations covered, and the time schedule for when participants were reporting.
Most of the surveillance systems studies were region specific, likely owing to their integration into regional public health networks and programmes. Seasonal influenza vaccine is the most frequent vaccine placed under this specific type of AEFI surveillance. AEFI surveillance conducted using digital solutions primarily relied on short message service (SMS) and/or email to interact with vaccinees and to send them e-questionnaires to report specific details regarding AEFI. Only one study used a phone app to both prompt participants to report and to collect data.
Reflecting on the findings of the review, the researchers note that, in order for active participant-centred reporting to be successful, there must be sufficient engagement from both those who have experienced an event and those who have not. Indeed, many of the studies identified in this review had response rates over 70%. Follow up prompts or phone calls for non-responders increased reporting rates. Fifteen studies in the review reported that any participant who reported a serious AEFI triggered human follow-up to assess the situation and collect additional information.
The researchers conclude that active, participant-centred, digital AEFI surveillance could become a primary method of collecting self-reported subjective symptoms and reactogenicity from vaccinees, complementing existing systems. However, future evaluation and study of these systems is essential, as is international expert collaboration, to identify novel applications and necessary improvements to match both contemporary populations and vaccine programmes. In that vein, the researchers are conducting a subsequent COVID-19-immunisation-surveillance-specific scoping review to describe a niche and evolving area of the research landscape.
Vaccine Volume 40, Issue 31, 29 July 2022, Pages 4065-4080. Image credit: SmartVax
- Log in to post comments












































